Melting #2: Copper (II) Sulfate (Didn’t Work)
The last time we melted sulfur, and it was really fun (except for cleaning the test tube). Now let’s melt something else, what about Copper Sulfate?
Copper (II) sulfate is the inorganic compound with the chemical formula CuSO4. Older names for this compound include blue vitriol, bluestone, vitriol of copper, and Roman vitriol. The pentahydrate (CuSO4·5H2O), the most commonly encountered salt, is bright blue.
Melting Point: 110 °C (230 °F)
Sulfur’s melting point is 5 °C higher (which means they’ll melt about the same time).
It looks impossible to melt it because the sulfur is more (soft) like a powder, but this one is tiny crystals. Let’s give it a try anyway.

Light the lamp!

OK, it’s heating it up nicely.
A couple minutes later:

The copper sulfate is turning whiter, but still, all of it still remains solid.
But look. There’s water vapor in there. That’s weird, maybe there’s too much heat? But the sulfate didn’t melt yet.
10 minutes later:

The sulfur melted already at this time. But the sulfate still remains a solid and it’s just turning whiter.
20 minutes later:
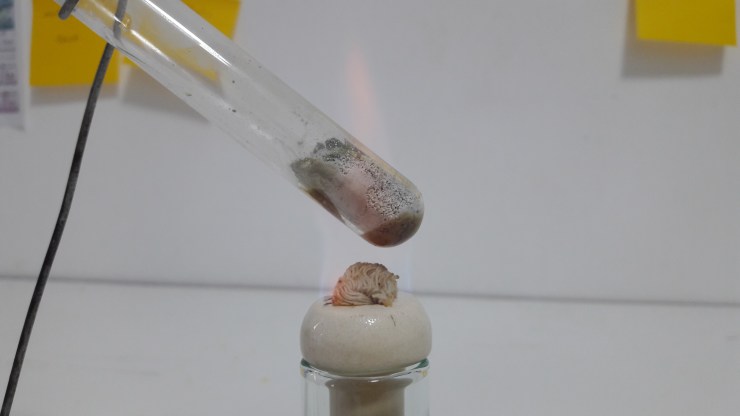
This is taking forever! It’s not melting. Did I do something wrong?

It’s 115 °C already, and the temperature can go further.
I guess it won’t melt anymore so I turned off the heat.
Wow! this experiment is a fail. I wonder why it has water vapors? Why is it turning white? When is it actually going to turn to liquid?
Any ideas why it didn’t melt? Feel free to comment down below ↓


You’re welcome 😊💐
You’re the best! Thank you so much!! I really appreciate what you did! 😆😆😆
Reblogged this on Creative Crafts DIY and commented:
Mr little einstein 👌😊
“110 °C (230 °F; 383 K) decomposes (·5H2O) <560 °C decomposes" Found this on Wikipedia, not sure what it means but I'll find out.