What is Silica Gel?
Silica gel is a granular, vitreous, porous form of silicon dioxide made synthetically from sodium silicate. Silica gel contains a nano-porous silica micro-structure, suspended inside a liquid. Most applications of silica gel require it to be dried, in which case it is called silica xerogel. For practical purposes, silica gel is often interchangeable with silica xerogel. Silica xerogel is tough and hard; it is more solid than common household gels like gelatin or agar. It is a naturally occurring mineral that is purified and processed into either granular or beaded form. As a desiccant, it has an average pore size of 2.4 nanometers and has a strong affinity for water molecules.
This is how it looks like (I got this one from a medicine bottle):
Silica gel is most commonly encountered in everyday life as beads in a small (typically 2 x 3 cm) paper packet. In this form, it is used as a desiccant to control local humidity to avoid spoilage or degradation of some goods. Because silica gel can have added chemical indicators and absorbs moisture very well, silica gel packets usually bear warnings for the user not to eat the contents.
Because Silica gel is non-toxic, non-flammable, and non-reactive and stable with ordinary usage. It will react with hydrogen fluoride, fluorine, oxygen difluoride, chlorine trifluoride, strong acids, strong bases, and oxidizers. Silica gel is irritating to the respiratory tract and may cause irritation of the digestive tract, and dust from the beads may cause irritation to the skin and eyes, so precautions should be taken.
Silica gel’s high specific surface area (around 800 m2/g) allows it to absorb water readily, making it useful as a desiccant (drying agent). Silica gel is often described as “absorbing” moisture, which may be appropriate when the gel’s microscopic structure is ignored, as in silica gel packs or other products.
An aqueous solution of sodium silicate is acidified to produce a gelatinous precipitate that is washed, then dehydrated to produce colorless silica gel. When a visible indication of the moisture content of the silica gel is required, ammonium tetrachlorocobaltate(II) (NH4)2CoCl4 or cobalt chloride CoCl2 is added. This will cause the gel to be blue when dry and pink when hydrated. An alternative indicator is methyl violet which is orange when dry and green when hydrated. Due to the connection between cancer and cobalt chloride, it has been forbidden in Europe on silica gel.
Once saturated with water, the gel can be regenerated by heating it to 120 °C (250 °F) for 1–2 hours. Some types of silica gel will “pop” when exposed to enough water. This is caused by breakage of the silica spheres when contacting the water.
Now let’s play with it.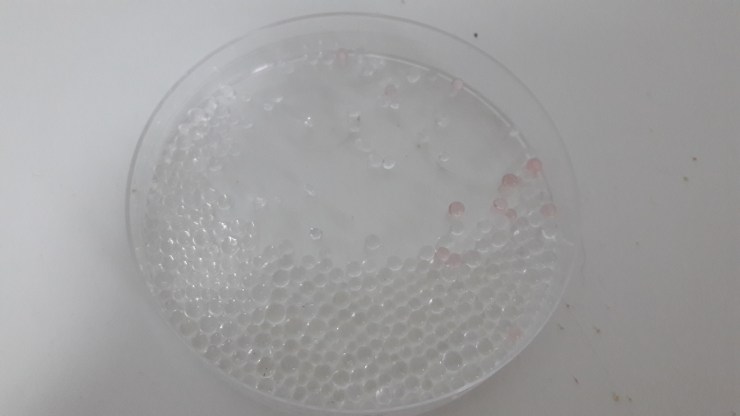
I got these from two packets, that’s why there’s so many.
I put about 5 ml of water into this little graduated cylinder. After that, I poured the gel in there and wait for a couple minutes.

Time to measure the water.

Look at that, the water went down about 1.4 ml. It actually absorbs water. I wish I could weigh the gel though.

Did you learn anything? If you did, drop a like below ↓
Sources:
Thank you so much for the nice comment
I use food grade packs in the dehydrated food I make. They are amazing! Great blog!
Yes – I find these packets in vitamin bottles etc And I always think they are superfluous. Now I know otherwise! 🙂
A scale is one of the things in science equipment that I can’t get. I have to order it from the internet… Anyway, I like these gels a lot, they’re very useful. It surprised me how much water it absorbs too.
I’m impressed that the silica gel absorbed so much water!
I use these types of little packets to keep my flash sticks dry where I keep them in a box.
And I agree with you – it would have been nice to weigh the gel. I think you will be adding ‘scales’ to your wish list! 🙂